The pressure p in atmospheres (atm) is equal to the pressure p in millimeter mercury (0°c) (mmHg) times 0.
#728 MMHG TO ATM HOW TO#
How to convert Millimeter Mercury (0☌) to Atmospheres 1 millimeter mercury (mmHg) is equal to 0.00132 atmospheres (atm). And so these are your final answers and the conversion factors can be found just searching on Google. 728.3 Millimeter Mercury (0☌) 0.95829 Atmospheres. These units canceled out and then work it. 103 3.3 Pascal's is equal toe one millimeter Mercury. So again these units cancel out, and this value is 194 Neiers mercury, and then the next one is given a millimeters mercury and once in Pascal's, so we need to multiply by the conversion factor. The absolute atmospheric pressure in the laboratory that day is 728 mm Hg. The volume of the flask is known to be 231.1 mL. the mercury in the arm attached to the gas is 13.6 cm higher than the one open to the atmostphere atmospheric pressure is 1.08. After it is cool, she momen- tarily removes the stopper, then replaces it and weighs the flask and condensed vapor, obtaining a mass of 53.780 g. What is the pressure of the enclosed gas in torr in each situation.
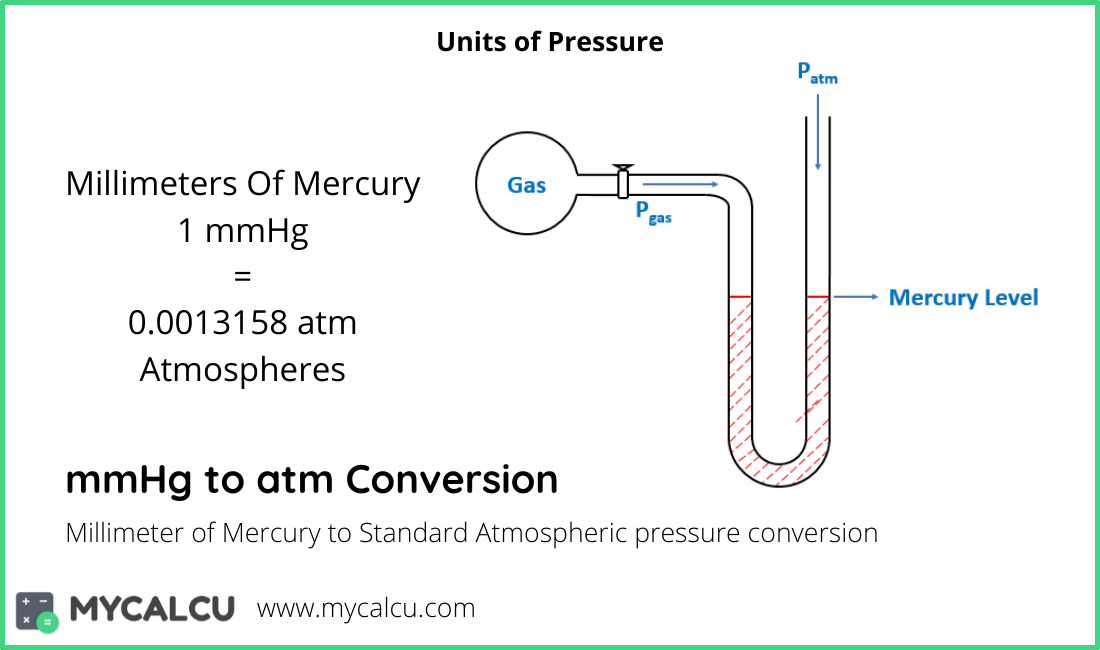
So this conversion factor is 760 millimetre. An open ended manometer containing mercury is connected to a container of gas. If the volume of a gas is 25.8 mL at 28.7☌ and 728 mmHg, what is its volume at 15.2☌ and 757 mmHg You should be able to do this calculation by reading the experimental write-up.

And then we can just do the same thing with the next ones. You can do it like this: 760'mmHg'1' ''Atm' .1'mmHg'1/760' ''Atm' .520'mmHg'1/(760)xx5200. Note: 1 atm 760 mm Hg where 760 is an exact number (not 2 sig. Is it? One killer passed out is equal to 7.5 tours because it's basically like you're multiplying by one so the units cancel out and then we get 46.93 Pascal's or killed US girls. So first you need to multiply by the conversion doctor whenever you're trying to get one unit to a different unit, so we need tours on the bottom for it to cancel out. So first it gives us 352 tours and then once that, and kill a pascal's. If 1 atm has to be converted to bar, $1atm\,=\,1.To this problem is just asking for basic dimensional analysis. The formula to convert from mmHg to atm is: atm mmHg ÷ 760. Let’s see some interconversion from 1 atm. Lets take a closer look at the conversion formula so that you can do these conversions yourself with a calculator or with an old-fashioned pencil and paper. Note: We know the interconversion between the units to do various calculation problems. So we can expresses the equation of pressure as, We know that pressure is the force exerted in a specific area and it is calculated by dividing the force applied by the area on which the force was applied. So before going into the solution part of the question, let’s discuss some concepts about pressure and its unit.
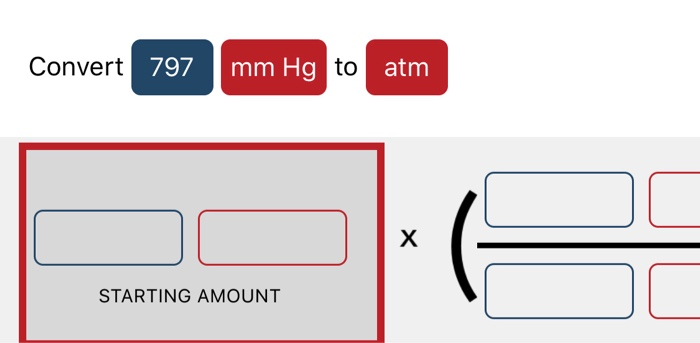

In the question, it is asked that we have to convert the given value of unit in mm Hg to atmospheres of pressure.The question is simply about te unit conversion of values between the units of pressure, We should be familiar with 1 atm value equal to how much value of mmHg. 728 Atmospheres 553279.99999 Millimeter Mercury (0☌) How to convert Atmospheres to Millimeter Mercury (0☌) 1 atmospheres (atm) is equal to 760 millimeter mercury (0°c) (mmHg). Hint: The approach for the question should be done by focusing on the unit conversion of the value from mmHg to atmospheres of pressure in atm.
